What is a Lithium Battery?
A Li-ion battery or Lithi-ion battery/ Lithium-ion battery, sometimes abbreviated as LIB, is a type of rechargeable battery. During the charging process, Li-ions move from the positive electrode to the negative electrode, and vice versa during the discharge process (usage process). LIBs often use electrodes that are compounds whose crystal structure has a layered form (layered structure compounds); in this case, during the charging and discharging processes, Li-ions will penetrate and fill the spaces between these layers, allowing the chemical reaction to occur. Common layered crystal structure electrode materials used for the positive electrode are compounds of transition metal oxides and Li, such as LiCoO2, LiMnO2, etc.; graphite is used for the negative electrode. The battery’s electrolyte solution allows Li-ions to move from one electrode to the other, meaning it has the ability to conduct Li-ions; however, the requirement is that this solution must not conduct electricity.
When discharging (usage process), the battery discharges through the external circuit, electrons from the anode (negative electrode) move to the cathode (positive electrode). Lithium ions move within the battery, also from the negative electrode to the positive electrode. When charging, under the charging voltage, electrons move to the anode (which at this time becomes the positive electrode); to balance the charge, inside the battery, Lithium ions move from the cathode (which at this time becomes the negative electrode) to the anode.

LIBs are often used for portable electrical devices, most commonly rechargeable batteries for handheld electronic devices. Li-ion batteries have a high energy density, a very small memory effect, and low self-discharge. Currently, in developed countries, LIBs are being focused on for development in the military, applications for electric vehicles, and aerospace engineering. It is expected to replace lead-acid batteries in cars, motorcycles, and electric vehicles. Furthermore, replacing lead-acid batteries also promises to ensure a clean environment and improve usage safety by avoiding the use of acid-containing electrolyte solutions, and limiting heavy metal emissions into the environment, while Li-ion batteries still ensure a voltage comparable to batteries.
Chemical composition, performance, cost, and safety are the basic factors that define the different types of LIBs. Handheld electrical devices (such as mobile phones, laptops) currently almost exclusively use LiCoO2 (abbreviated LCO) Lithium Cobalt Oxide as the negative electrode. This material has a high energy density but is less safe, especially dangerous when the battery leaks. Lithium iron phosphate (LiFePO4, or LFP), Lithium Manganese Oxide (LiMn2O4, Li2MnO3, or collectively LMO), and Lithium Nickel Manganese Cobalt Oxide (LiNiMnCoO2, or NMC) are other common positive electrode materials; however, they have lower energy density than LCO, but have a longer life cycle and are safer. Batteries using these materials are often used in medical electrical equipment. NMC, in particular, is currently a leading candidate for batteries used in electric vehicles. Lithium Nickel Cobalt Aluminum Oxide (LiNiCoAlO2 or NCA) and Lithium Titanate (Li4Ti5O12 or LTO) are used for special purposes. Lithium-sulfur batteries are a newly developed type, holding much promise due to their high performance and low mass.
See more: Capacity Profile Of Phuc Gia Laboratory Corporation
Because Lithium-ion batteries contain a flammable electrolyte, compressed under high pressure, they become particularly dangerous. If a battery is charged too quickly, it can cause a short circuit, leading to fire and explosion. Due to this risk, the testing standards for LIBs are much stricter than for acid electrolyte batteries. An example of a battery failure causing serious damage is the Samsung Galaxy Note 7 battery incident in 2016.
Research fields for lithium-ion batteries include increasing lifespan, energy density, safety, and reducing cost.
History of Lithium Battery Development
The Lithium Battery was developed by the British chemist M. Stanley Whittingham, currently teaching at Binghamton University, while he was working for Exxon in the 1970s. Whittingham used titanium (IV) sulfide and lithium metal as electrodes. However, this rechargeable lithium battery could never be put into practice. Titanium disulfide was a poor choice, as it had to be synthesized in a complete vacuum. This was extremely expensive (~$1,000 per kilogram of titanium disulfide in the 1970s). When exposed to air, titanium disulfide reacts to form hydrogen sulfide compounds with an unpleasant odor. For this and other reasons, Exxon discontinued Whittingham’s titanium disulfide-lithium battery. Batteries with lithium metal electrodes showed safety problems, as lithium is a highly reactive substance; it burns in normal atmospheric conditions due to water and oxygen in the air. Therefore, research shifted to developing batteries that do not use metallic lithium, but instead use chemical compounds of lithium, with the ability to accept and release lithium ions.
The Li-ion battery was first commercialized by Sony Energitech in 1991. Today, Li-ion batteries have become the dominant battery type in the market for mobile devices worldwide.
Operating Principle of the Lithium Battery
The reactants in the electrochemical reaction in a lithium-ion battery are the negative and positive electrode materials; the electrolyte solution provides a conductive medium for lithium ions to move between the two electrodes. Current flows in the external circuit when the battery is running.
Lithium ions move within both electrodes during the reaction. Most current electrode materials are materials that allow lithium ions to penetrate and enter the crystal lattice, with no or minimal disturbance to the positions of the remaining atoms in the lattice during the lithiation (intercalation/insertion) process, and conversely, lithium ions leave the crystal lattice (deintercalation/delithiation/extraction process).
When discharging, lithium ions (positively charged) move from the negative electrode (anode), usually graphite (C6 in the reaction below), through the electrolyte solution, to the positive electrode, where the positive electrode material will react with the lithium ions. To balance the charge between the 2 electrodes, for every Li ion that moves from the negative electrode to the positive electrode (cathode) inside the battery, one electron moves from the negative electrode to the positive electrode in the external circuit, meaning it generates a current flowing from the positive electrode to the negative electrode.
When charging, the reverse process occurs; under the charging voltage, electrons are forced to flow from the positive electrode of the battery (now becoming the negative electrode), Li ions detach from the positive electrode and move back to the negative electrode of the battery (now acting as the positive electrode). Thus, the battery reverses direction during the charging and discharging processes. The names positive or negative electrode must be determined based on the nature of the reaction and the process being monitored. In this article (and in most scientific papers), the negative electrode (anode) and positive electrode (cathode) of the battery are always named based on the discharge state.
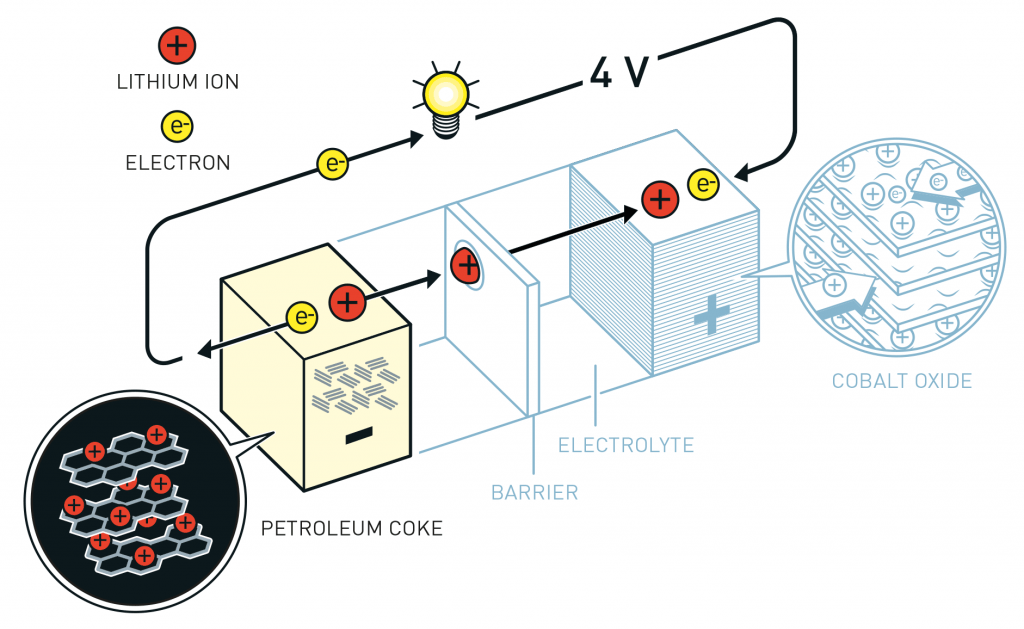
The half-reaction at the positive electrode (cathode) in the layered $LCO$ material is written as follows (forward direction is charging, reverse direction is discharging):
LiCoO2 ↔ CoO2 + Li+ + e–
The half-reaction at the negative electrode (anode) in the layered graphite material (forward direction is charging, reverse direction is discharging):
C6 + Li+ + e– ↔ LiC6
The reaction of the whole battery (forward direction is charging, reverse direction is discharging)
C6 + LiCoO2 ↔ LiC6 + CoO2
Thus, when charging, C60 (anode) is reduced to C61-, Co3+ is oxidized to Co4+, and vice versa when discharging.
Basically, the reactions always have limits. If over-discharging (stuffing excess Lithium ions) a saturated Lithium Cobalt Oxide, it will lead to the formation of Lithium Oxide, according to the following one-way reaction:
LiCoO2 + Li+ + e– → Li2O + CoO
If overcharging the $LCO$ battery potential above 5.2 V, it will lead to the formation of Cobalt (IV) Oxide, according to the following one-way reaction; this has been verified by X-ray diffraction.
LiCoO2 → Li+ +e– + CoO2
Structure of a Lithium Battery
Positive electrode (cathode)
The material used for the positive electrode is usually from LiCoO2 and LiMnO4. Cobalt-based materials often have a pseudo-tetrahedral structure, allowing lithium ions to diffuse in 2 dimensions. These are ideal materials capable of providing high specific power, high volumetric specific power, limited self-discharge, high potential, and long cycle life. Its limitations are high cost due to containing cobalt, a rare metal, and poor thermal stability. Manganese-based materials have a cubic crystal system, allowing lithium ions to diffuse in all three dimensions. This material is gaining interest because manganese is cheaper and more abundant than cobalt, has higher performance, and a longer cycle life, if its other limitations are overcome. These limitations include the solubility of the manganese material in the electrolyte solution, making the electrode less durable and reducing battery capacity. Cobalt-containing positive electrode materials are the most common type; however, other materials are currently being researched to lower costs and increase battery capacity. As of 2017, LiFePO4 is expected to bring high applicability for large-sized batteries, such as those used for electric vehicles, due to its low cost and high power, although this material has low conductivity and the use of carbon as a conductive additive is mandatory.
| Positive electrode | ||||
| Compound | Corporation | Application | Year | Advantages |
| Lithium Nickel Manganese Cobalt Oxide (NMC, LiNixMnyCozO2) | Imara Corporation, Nissan Motor, Microvast Inc., LG Chem | Electric vehicles, power tools, grid energy storage | 2008 | High specific energy and specific energy density |
| Lithium Manganese Oxide (LMO, LiMn2O4) | LG Chem, NEC, Samsung, Hitachi, Nissan/AESC, EnerDel | Hybrid electric vehicles, phones, laptops | 1996 | Low cost, durable, high specific energy |
| Lithium Iron Phosphate (“LFP”, LiFePO4) | University of Texas/Hydro-Québec, Phostech Lithium Inc., Valence Technology, A123Systems/MIT | Segway Personal Transporter, power tools, aerospace products, automotive hybrid systems, PHEV conversions | 1996 | Medium energy density (2 A·h outputs 70 amperes) Safe, thermally stable |
| Lithium Cobalt Oxide (LiCoO2) | Sony first commercial production | Diverse | 1991 | High specific energy |
| Lithium Nickel Cobalt Aluminum Oxide (“NCA”, LiNiCoAlO2) | Panasonic, Saft Groupe S.A. | Electric vehicles | 1999 | High specific energy, long cycle life |
Negative electrode (anode)
The commonly used anode material is graphite and other carbon materials. They are very cheap and abundant, as well as having good electrical conductivity and a structure that allows lithium ions to intercalate between the layers in the carbon lattice, thereby being able to store energy while the crystal structure can swell up to 10%. Silicon is also used as an anode material because it can also hold lithium ions, even more than carbon; however, when “holding” lithium ions, silicon can swell to over 400% of its original volume, thus destroying the battery structure.
| Negative electrode | |||||
| Compound | Storage | Corporation | Application | Year | Comment |
| Graphite | 372 mAh/g | Is the main material for the negative electrode in most LIBs | 1991 | Low cost. Charging speed depends heavily on the structure, size, and shape of each graphene layer. | |
| Lithium Titanate (“LTO”, Li4Ti5O12) | 175 mAh/g | Toshiba, Altairnano | Automobiles (Phoenix Motorcars), grid energy storage (PJM Interconnection Regional Transmission Organization control area, United States Department of Defense), bus (Proterra) | 2008 | Current, charging time, durability (safe, thermally stable, can run in the range −50–70 °C (−58–158 °F)) |
| Hard Carbon | 540 mAh/g | Energ2 | Household power tools | 2013 | Large capacity |
| Tin cobalt alloy (CoSnx) | Up to 992 mAh/g | Sony | Power tools (Sony Nexelion battery) | 2005 | Larger capacity than graphite battery (3.5Ah 18650-type battery) |
| Silicon/Carbon | Volumetric: 580 W·h/l | Amprius | Smartphones, with 5000 mA·h capacity | 2013 | Requires a nano structure with silicon content <10% by mass |
Silicon can be used as a negative electrode material; however, its reaction with Li can cause the material to fracture. This fracture exposes the inner Si layers directly to the electrolyte solution, so they can decompose, forming a solid electrolyte interphase (SEI) layer on the newly formed Si surface. This SEI layer can thicken, preventing the diffusion of Li+ and reducing the electrode’s capacity as well as battery power and decreasing the anode’s durability. Many efforts are being made to minimize the structural changes due to Si fracture, such as synthesizing Si in the form of nanofibers, nanotubes, hollow spheres, nanoparticles, and porous nano structures.
Electrolyte solution (electrolyte)
The electrolyte solution or electrolyte is the medium for lithium ion transport between the electrodes during the battery’s charging and discharging processes. Therefore, the basic principle for an electrolyte solution for Li-ion batteries is that it must have good ionic conductivity, specifically a lithium ionic conductivity of 10−2 S/cm at room temperature, increasing by about 30-40% when up to 40°C and decreasing slightly when the temperature drops to 0°C. During the battery’s charging and discharging, as lithium ions move within the battery, leading to a potential difference, the battery generates a current in the external circuit where electrons transfer from the negative to the positive electrode (always in the same direction as the lithium ions). To ensure the reaction occurs in the battery and the battery does not short-circuit, the electrolyte solution needs to be a good insulator, meaning the electron conductivity of this solution must be at or below 10−8 S/cm. Liquid electrolyte solutions used in Li-ion batteries contain lithium salts, such as LiPF6, LiBF4 or LiClO4 in an organic solvent such as ethylene carbonate, dimethyl carbonate, and diethyl carbonate.

Because organic solvents often decompose easily at the negative electrode during charging, during the first charge, a solid electrolyte interphase (SEI) layer often forms at the negative electrode, which can reduce the anode’s conductivity. This interphase layer can prevent the decomposition of the electrolyte solution, and thereby form a stable interface layer.
Composite electrolyte solutions based on an organic polymer (poly(oxyethylene)) can also be a stable interface layer. It can be used to coat the electrode surface for protection in Li-polymer batteries, or in other normal li-ion batteries.
To limit the leakage of electrolyte solutions with organic solvents, and to increase safety as well as minimize flammability when this solvent encounters air, gel solvents, polymers, or ceramic-based solid-state electrolytes are being actively developed.
When using a Solid Electrolyte, a solid-state li-ion battery is obtained; in this case, the separator film can be eliminated, simplifying the assembly process and increasing safety for the battery.
Charging and Discharging Mechanism of a Lithium Battery
The process of regulating the charge/discharge of a single Li-ion battery cell and a complete Li-ion battery system, which includes multiple battery cells connected in series, is relatively different.
A single Li-ion battery cell is charged/discharged in two stages:
1 – Constant current (CC) mode
2 – Constant voltage (CV)
For a complete Li-ion battery system, there’re 3 stages required:
1 – Constant current (CC)
2 – Balance
3 – Constant voltage (CV)
In the constant current mode, the charger will apply a constant current to the battery through a steadily increasing voltage until the battery’s cutoff voltage is reached. In the balancing mode, the charger gradually reduces the charging current to the battery, or regulates by turning the charging current on and off so that the state of charge for each battery cell reaches a balanced state within the circuit, until all cells in the circuit are balanced. Some charging devices regulate balance by charging each battery cell sequentially; however, this prolongs the charging time. Creating an algorithm to optimize this balancing process can increase performance and optimize battery charging time. In the constant voltage mode, the charger applies a voltage equal to the cutoff voltage of each cell multiplied by the number of cells connected in series to the entire battery; this is essentially the discharge process, so the current will decrease to 0, until the current is below the 3% threshold of the initial charging current value, then the battery stops operating. If charging/discharging exceeds the allowable potential and current thresholds, it can lead to battery explosion.

Operating Temperature of a Lithium Battery
The temperature limit of the battery when charging is more important than the discharge temperature (temperature during use). Scientists have found that it is running at too high a temperature (not for too long) that reduces battery life. The battery will work best when charged at 5-45°C; at this time, high-speed charging is possible. At lower temperatures, i.e., 0-5°C, it can be charged but the current will decrease, although during charging, the battery’s temperature will increase slightly due to the battery’s internal resistance. The phenomenon of temperature increase during charging is the cause of reduced battery performance; when the temperature rises above 45°C, the battery will degrade quickly. However, when charging at low temperatures, the battery’s internal resistance increases, reducing the speed and increasing the charging time.
LIB batteries should not be charged at temperatures below 0°C. At this temperature, although the battery system appears to be charging normally, due to the low temperature, the poor conductivity of the electrode materials will reduce the reactivity of lithium ions with the electrode material. In that case, lithium will be plated onto the electrode surface instead of diffusing deep into the material and participating in the reaction under cold charging conditions; this plated layer adheres tightly to the electrode even if charging or discharging continues. Therefore, most batteries cannot operate outside the 0-45°C range due to safety factors.
ARTICLES RELATED TO REGULATION CONFORMITY SERVICES:
For more details, please contact us at:
PHUC GIA LABORATORY CORPORATION
PHUC GIA CERTIFICATION CENTER
PHUC GIA INSPECTION & TESTING CENTER
Address: ICD Long Bien, No. 01 Huynh Tan Phat, Sai Dong B Industrial Park, Long Bien Ward, Hanoi City, Vietnam.
Hotline: 0981 996 996/ 0982 996 696/ 024 7779 6696
E-mail: lab@phucgia.com.vn/cert@phucgia.com.vn/info@phucgia.com.vn
Website: phucgia.com.vn
Working time: Monday to Friday 8:00 – 18:30; Saturday 8:00 – 12:00


![QUOTATION – Testing and Other Certification Services [2026]](https://phucgia.com.vn/wp-content/uploads/2017/03/bao-gia-thu-nghiem-chung-nhan.png)





