Phuc Gia Certification Center (PGL) has compiled the Process for Certification for IP Cameras (Surveillance cameras using Internet Protocol) compliant with QCVN 135:2024/BTTTT at Phuc Gia®.
See more: Capacity Profile Of Phuc Gia Laboratory Corporation
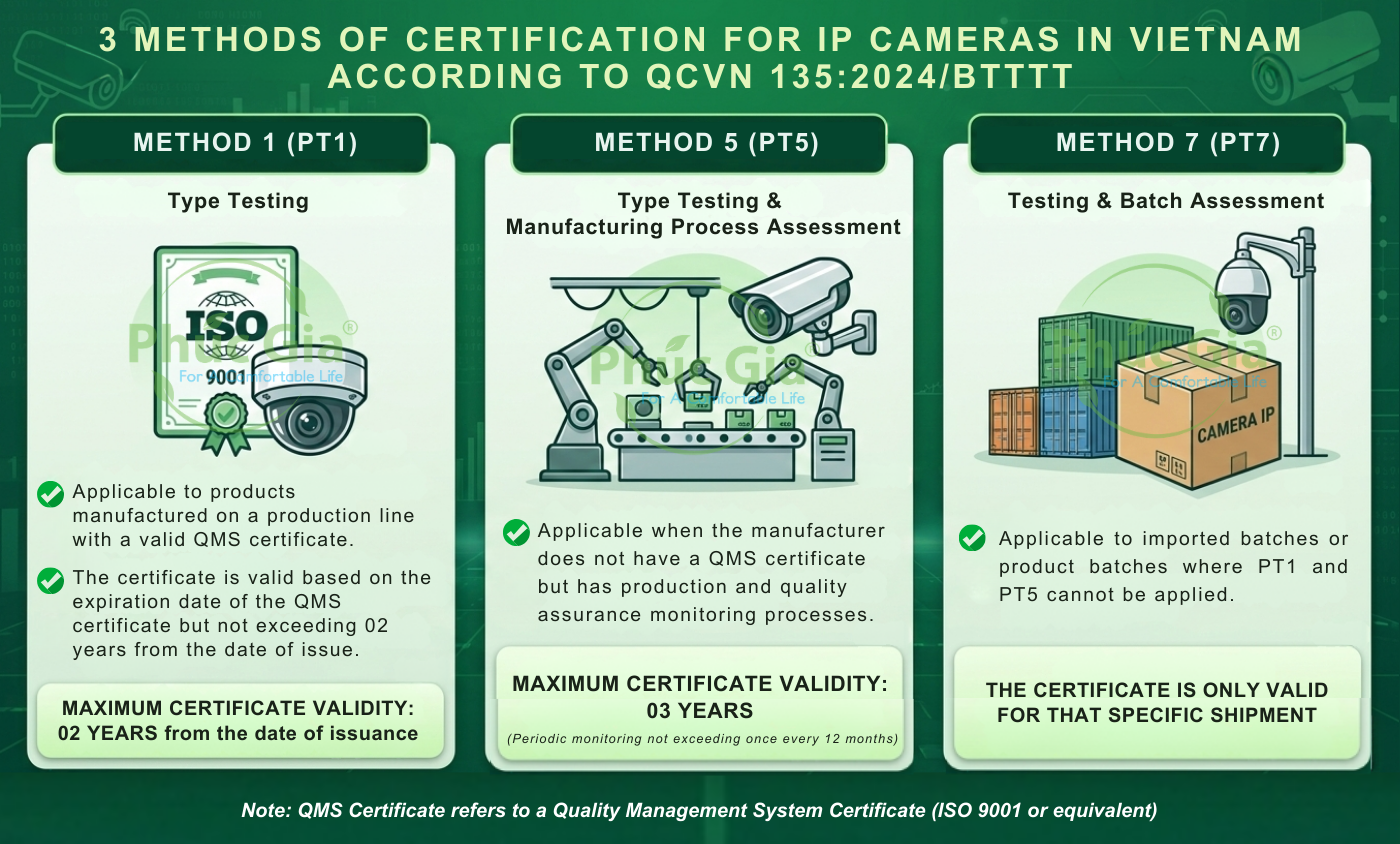
I. Appropriate Certification Methods
IP Camera products are certified using one of the following three methods:
II. Implementation Process for Certification for IP Cameras in Vietnam compliant with QCVN 135:2024/BTTTT
Step 1. Receipt of Certification Request
Customers submit dossiers to PGL for certification including:
- Certification registration form according to the template:
- Form PGL-TT21-BM01a for Method 1 or Method 7;
- Form PGL-TT21-BM01b for Method 5.
- IXIT documents;
- Firmware;
- Goods labels (Main labels, sub-labels if any) compliant with QCVN 135:2024/BTTTT, Decree No. 37:2026/ND-CP, and current legal regulations;
- Copy of Business Registration Certificate or Investment Certificate (latest version);
- Product technical documents;
- Product user manuals;
- Method 1: Quality Management System Certificate (ISO 9001 or equivalent) with a scope of Camera manufacturing and still valid (minimum 3 months) in English or Vietnamese;
- Method 5: Quality management document system (such as Quality Manual, production processes, quality control plans…);
- Method 7: Import dossier of the shipment (Contract, Invoice, Customs Declaration, Packing List, Bill of Lading, …) or Production dossier (for domestic production batches);
- Other documents (if any).
SPECIAL NOTE:
- If IXIT documents or Firmware are missing (or incomplete), the dossier will be considered NON-CONFORMING and will NOT be accepted for processing.
- Conforming dossiers will be received and processed after the Customer signs the contract and performs the obligations specified in the contract.
Step 2. Dossier Review and Processing Time
1. PGL reviews the dossier:
- Check the consistency of information, product technical characteristics… and the registration dossier;
- Review the certification method: Check the suitability of the applied method (1, 5, or 7);
- For Method 1 (PT1): Review the suitability of the manufacturer’s Quality Management System Certificate provided by the customer.
2. Dossier processing time:
- PT1 and PT7: No more than 05 working days from the time the dossier is complete and valid (excluding the time for field inspection, sampling, and testing).
- PT5: Assessment time depends on the production scale, number of models, and actual sampling lot.
3. Establish an assessment team:
The PGL assessment team will include:
- Assessor;
- Technical expert (if any).
Step 3. Field Inspection and Sampling
- PGL will directly conduct a field inspection (PT1, PT5, PT7): Check the appearance and compliance of labeling (based on the Registration Dossier, QCVN 135:2024/BTTTT, Decree No. 37/2026/ND-CP, and current regulations…);
- PGL will directly collect samples for certification: Collect 04 products for each model (including 02 samples for testing and 02 samples for storage).
Note:
- Samples stored at the enterprise: The enterprise must ensure storage conditions, and only Phuc Gia® is allowed to break the seal during the sample storage period.
- Sample storage time: Within 02 years (for PT1 and PT7) / 03 years (for PT5) from the date of issuance of the Certificate.
- In case the sample storage period has not expired and the enterprise wishes to use them, they must send a written request to Phuc Gia® and commit to taking responsibility for subsequent issues.
Step 4. Testing and Evaluation of Results
1. In case the test samples PASS:
PGL will officially issue the Certificate and the Conformity Certification Mark to the customer (Step 5).
2. In case the test samples FAIL:
– For Method 1 (PT1): PGL notifies the “Failed” certification result.
- If the customer performs corrections and re-registers for certification, it shall be carried out as the initial procedure (Some dossiers previously provided by the customer may be used).
- In case the customer has a written request for testing the stored samples, Phuc Gia® will re-test the stored samples.
– For Method 5 (PT5): PGL notifies the “Failed” certification result and requests the customer to determine whether to continue testing the stored samples or not.
- If yes, proceed to transfer the stored samples to the laboratory, and the test results of the stored samples shall be the basis for concluding the certification result.
- If the Customer needs a second certification (after correcting and improving the product) for the above products, they shall establish a re-registration dossier from the beginning according to current regulations (the second assessment time must not be more than 60 days later than the first assessment time).
- If not, the certification ends.
– For Method 7 (PT7): PGL notifies the “Failed” certification result and requests the customer to determine whether to continue testing the stored samples or not.
- If yes, proceed to transfer the stored samples to the laboratory, and the test results of the stored samples shall be the basis for concluding the certification result.
- If the Customer needs a second certification (after correcting all cameras in the shipment), they are guided to establish a re-registration dossier from the beginning according to current regulations.
- If not, the certification ends.
Note:
- The customer is responsible for paying the testing fees for stored samples.
- The laboratory is approved by Phuc Gia® according to regulations.
Step 5. Dossier Review and Issuance of Certification Results
When the dossier is valid and the testing results of the IP Camera samples meet the requirements, PGL will officially issue the Certificate and the Conformity Certification Mark to the customer.
Note: Post-certification surveillance, re-certification, and expansion of certification scope activities are performed according to the Certification Management Process of Phuc Gia®.
III. Most Important Notes for New Customers
- Lack of Firmware and IXIT documentation is the leading cause for dossiers being rejected right from the receipt round. Please fully prepare these 2 documents.
- Proactively and carefully check appearance and labels: Before inviting Phuc Gia® to collect samples, customers need to ensure the shipment is intact, the packaging and labels are complete and correct according to the registered certification information, as well as ensuring compliance with current regulations (QCVN 135:2024/BTTTT, Decree 37/2026/ND-CP, …).
- Prepare a sufficient number of samples: At least 04 cameras must always be reserved for each model to serve both testing and sample storage. Breaking the seal of stored samples during storage at the enterprise is not allowed.
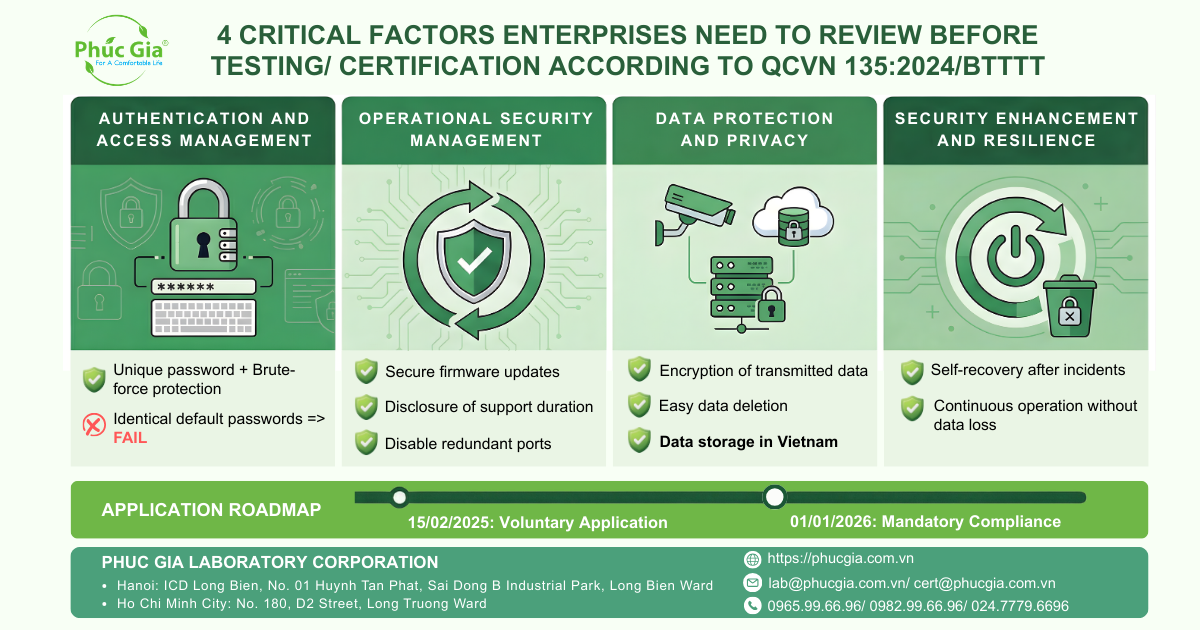
- Review and check to ensure 4 important factors (frequently failed) of the camera before registering for certification, including: Authentication and access management; Operational security management; Data protection and privacy; Security enhancement and resilience.
RELATED POSTS
- Phuc Gia® Designated to Certify Internet Protocol Surveillance Camera Equipment
- What is an IP Camera? The Role of IP Cameras in Daily Life
- Which Surveillance Cameras Must Undergo Testing According to QCVN 135:2024/BTTTT?
- The Impact of QCVN 135:2024/BTTTT on the IP Camera Market in Vietnam
- QCVN 135:2024/BTTTT on Basic Information Security Requirements for Internet Protocol Surveillance Cameras
- The Current State of Information Security Management for Surveillance Cameras Globally and in Vietnam
- Ensuring Network Information Security for Surveillance Camera Systems
For more details, please contact us at:
PHUC GIA LABORATORY CORPORATION
PHUC GIA CERTIFICATION CENTER
PHUC GIA INSPECTION TESTING CENTER
Address:
- Hanoi: ICD Long Bien, No. 01 Huynh Tan Phat, Sai Dong B Industrial Park, Long Bien Ward
- Ho Chi Minh City: No. 180, D2 Street, Long Truong Ward
Hotline: 0965996696 / 0982996696 / 02477796696
E-mail: lab@phucgia.com.vn/cert@phucgia.com.vn/info@phucgia.com.vn
Website: phucgia.com.vn
Working time: Monday to Friday 8:00 – 18:30; Saturday 8:00 – 12:00


![QUOTATION – Testing and Other Certification Services [2026]](https://phucgia.com.vn/wp-content/uploads/2017/03/bao-gia-thu-nghiem-chung-nhan.png)





